Double-Blind Randomized Clinical Trial: Gluten versus Placebo Rechallenge in Patients with Lymphocytic Enteritis and Suspected Celiac Disease | PLOS ONE

Evaluating Responses to Gluten Challenge: A Randomized, Double-Blind, 2-Dose Gluten Challenge Trial - Gastroenterology

Beyond Celiac - For an update on recent celiac disease research and progress on a treatment toward a cure, tune in to the latest episode of our podcast Celiac Straight Talk! Two

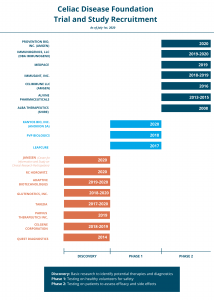
Celiac Disease Foundation on Twitter: "What happens during each phase of a clinical trial? Learn more about clinical trials and potential future therapies for celiac disease here. https://t.co/AytoKOhCIc https://t.co/c0hl5r4W4x" / Twitter

ImmusanT Publishes Positive Data from Phase 1 Trials of its Celiac Disease Drug Nexvax2 - Drug Discovery and Development









![Top 10 Celiac Disease Clinical Trials [2022 Studies] | Power Top 10 Celiac Disease Clinical Trials [2022 Studies] | Power](https://pwr-og-img.withpower.com/Apply%20to%20this%20Phase%202%20clinical%20trial%20treating%20Celiac%20Disease:%0A%0ATreatment%20for%20Celiac%20Disease.png?md=1)

![Top 10 Celiac Disease Clinical Trials [2022 Studies] | Power Top 10 Celiac Disease Clinical Trials [2022 Studies] | Power](https://pwr-og-img.withpower.com/Apply%20to%20this%20Phase%202%20clinical%20trial%20treating%20Fibrostenotic%20Crohn%C2%B4s%20Disease,%20Crohn%20Disease:%0A%0ASpesolimab%20for%20Fibrostenotic%20Crohn%C2%B4s%20Disease.png?md=1)
![Top 10 Celiac Disease Clinical Trials [2022 Studies] | Power Top 10 Celiac Disease Clinical Trials [2022 Studies] | Power](https://pwr-og-img.withpower.com/Apply%20to%20this%20clinical%20trial%20treating%20Celiac%20Disease%20in%20Children,%20Gastrointestinal%20Diseases,%20Digestive%20System%20Diseases,%20Intestinal%20Diseases,%20Gluten%20Sensitivity,%20Malabsorption%20Syndromes,%20Celiac%20Disease:%0A%0AGluten%20Powder%20for%20Celiac%20Disease%20in%20Children.png?md=1)

